The Backbone of Safety and Quality in Medical Devices
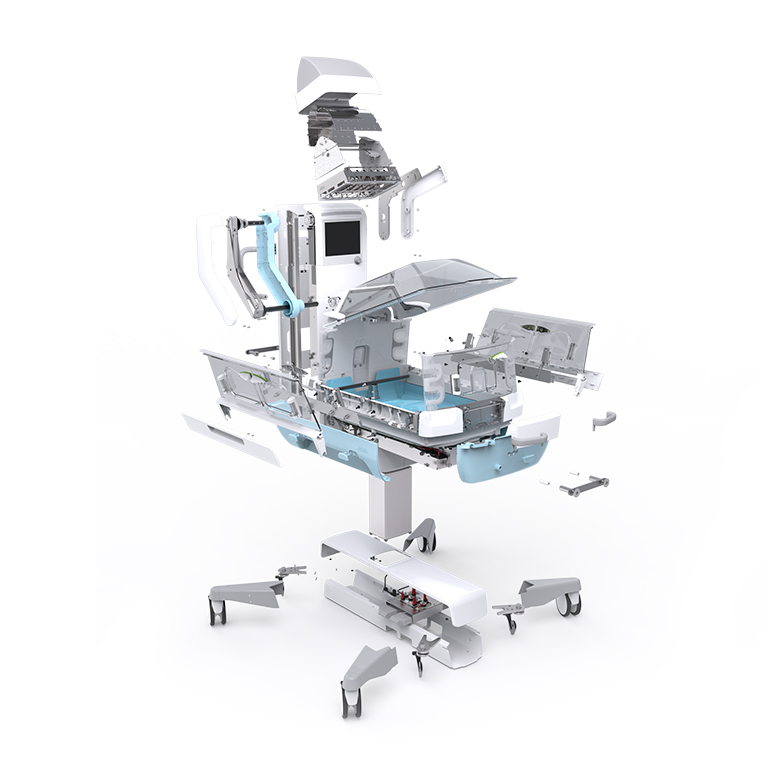
The medical industry is a place where innovation and trust are both extremely important. Every day, millions of individuals rely on an array of medical devices to monitor, diagnose, and treat various health conditions. From simple thermometers to complex cardiac monitors, these devices play a crucial role in ensuring the health and well-being of communities worldwide. The essence of trust in this domain goes from the assurance that these medical devices are designed with precision, operate with accuracy, and uphold an uncompromising standard of quality and safety. As the industry continually evolves to meet the dynamic healthcare demands, maintaining a stringent standard of quality and safety in medical devices is not just about meeting regulatory benchmarks; it’s about saving lives and enhancing the quality of healthcare. With this responsibility, comes the need for a solid framework that ensures safety and quality. This is where ISO 13485 jumps in.